|
5/20/2023 0 Comments Affinity chromatography
Currently studied affinity interactions are centered on lectins, antibodies and chemical analoges of enzymatic substrates. 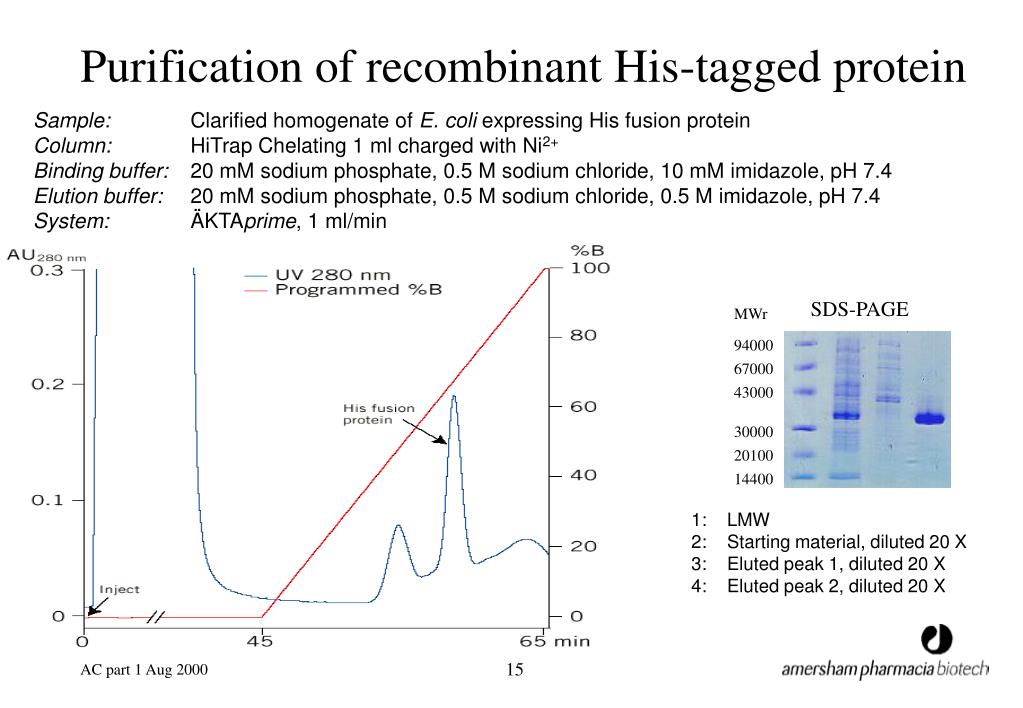
In our laboratory we are designing and optimizing different affinity matrices for the downstream processing of cell culture based (Madin Darby canine kidney cells) influenza vaccines. The interaction is based on structural properties, such as the interaction of antibodies with antigens, lectins with carbohydrates, enzymes with substrate analoges, hormones with receptors, and nucleic acids with binding proteins. proteins to immobilized molecules on solid support matrices based on the exploitation of known physico-chemical and biological affinities between the interacting molecules. Usually, they cannot use affinity chromatography to collect new or strange molecules.Affinity chromatography is a method of selectively and reversibly binding target compounds e.g. Sometimes the scientists must purify the target molecule to make sure they did not accidentally collect other molecules too.īecause scientists need to prepare a stationary phase with exactly the right ligand, they can only use affinity chromatography to collect molecules that they already understand well. Because most molecules from living things change shape depending on how much acid is nearby, the binding buffer and elution buffer can make the ligand and target molecule change shape to hold on tighter or let go of each other.Īffinity chromatography does not always work perfectly. That means they make it more acidic or less acidic. In some affinity chromatography, the binding buffer and elution buffer work by changing the pH of the solution. Scientists sometimes use machines or special tools to hold the stationary phase in exactly the right position and to pour the mixture, rinse, and other treatments in exactly the right way. Then the scientists add an elution buffer to make the ligand let go of the target molecule so they can collect it. The target molecule is stuck to its ligand, which is stuck to the stationary phase, so it is not rinsed away. Then the scientists rinse away the rest of the mixture. Sometimes, the scientists add a binding buffer to help the target molecule stick to its ligand. Then the scientists pour the mixture, or mobile phase over the stationary phase. The stationary phase can be membranes, special beads, or supports with different-sized pores in them. 
Then they prepare a stationary phase and treat it with the ligand. The ligand could be an antibody if the target molecule has an antigen DNA if the target molecule is a polymerase, DNA-binding protein, or DNA-binding enzyme aromatic molecules if the target is a different kind of enzyme or a peptide if the target is another biomolecule. 
To prepare for affinity chromatography, scientists decide which molecule they want and find its ligand. In affinity chromatography, the thing that the target molecule sticks to is called the ligand. A DNA-splicing protein will stick to its own DNA sequence but not to a different DNA sequence. A protein will stick to its own receptor or inhibitor but not to a different protein's receptor or inhibitor. An antibody will stick to its antigen but not to a different antibody's antigen. This laboratory technician has built an apparatus to perform affinity chromatography on a protein.Īffinity chromatography works because most molecules from inside living things stick to some molecules but not others, the way magnets stick to opposite magnets but not to wood or plastic.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
